 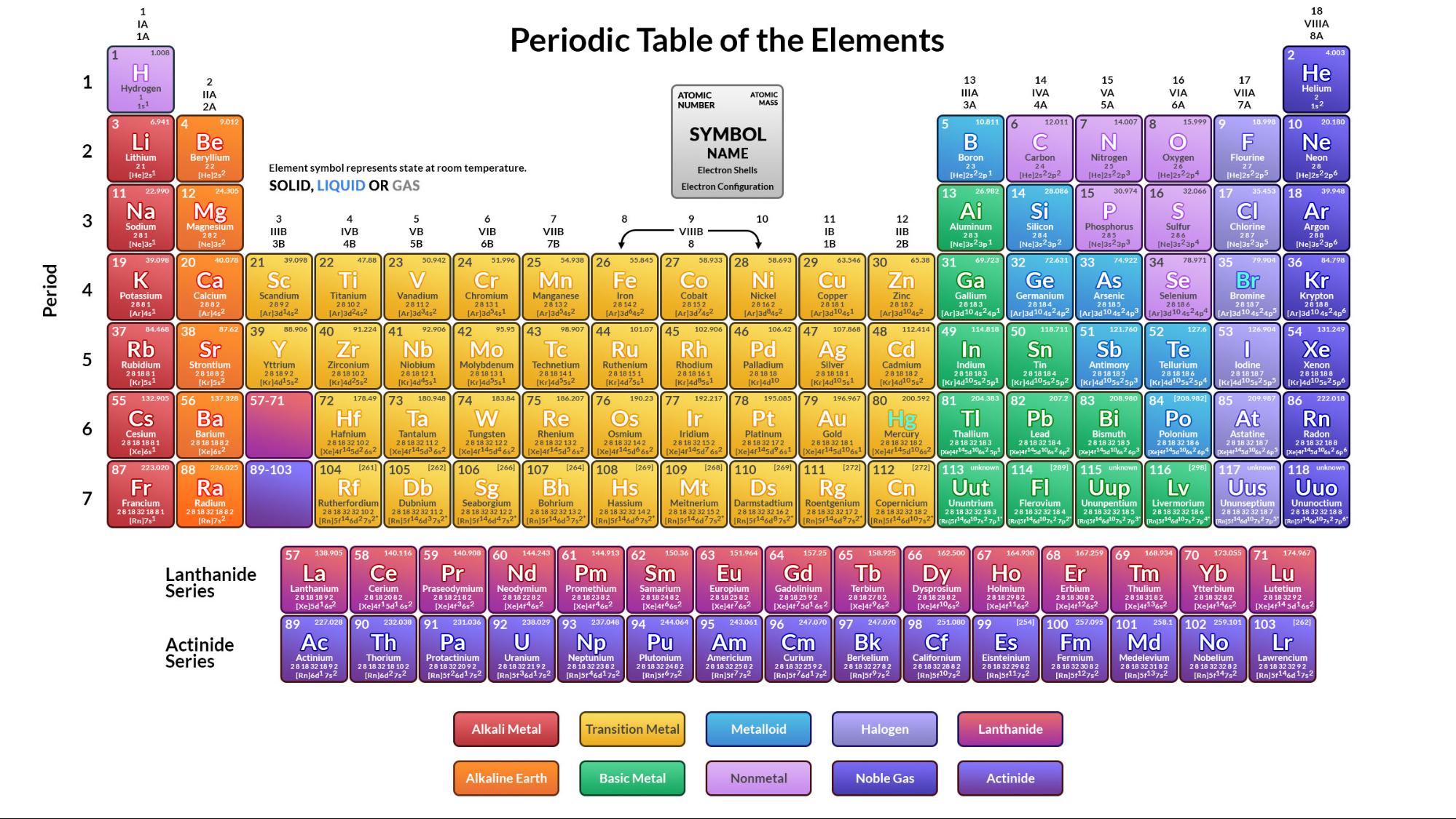
The numbers 1 to 18 refer to the different groupings.The periodic table consists of 18 groupings.The contemporary or extended form of a periodic table has vertical columns called groups. The number of the shells or energy levels in an atom of that element is represented by each period.The actinides are a group of 14 elements there in the seventh period. A separate section at the bottom of the lengthy version of the periodic table.With 32 components, this addition brings the 7th phase to a close. Four new elements respectively 113,115,117 and 118 have been added to the periodic table’s seventh period.The 6th period, on the other end, has 32 components. 
The fourth and fifth gained 18 elements per period.Each of the second and the third periods has eight components.The initial era is made up of only two elements: hydrogen and helium.They are labelled 1 to 7 from top to bottom.In the contemporary or long form of such periodic tables, periods are just horizontal rows. And it was observed that elements with comparable qualities recur at regular intervals. Across each row, scientists placed elements in ascending order of their atomic number, and this order is from left to right. The physical properties and chemical properties of elements are periodic functions of their atomic numbers, according to the contemporary periodic law. Relationships were more easily detected among compounds than among elements, resulting in an element categorization that was several years behind other compounds.įor over half a century, no widespread consensus on the categorization of elements had been obtained among chemists, even though compound classification methods had become widely used. This fast increase of chemical knowledge demanded categorization because not only the formalised literature of chemistry but also the laboratory techniques through which chemistry has been passed down as a living science through one generation of chemists are founded on classification. History:Ĭhemical analysis is an art of discriminating between distinct chemical substances that underwent fast growth in the early nineteenth century, resulting in the accumulation of a substantial body of information about the chemical characteristics and physical characteristics of both elements or compounds. In short, The periodic table is a diagram that depicts all of the elements with their physical properties, and it is arranged by atomic numbers & electron configurations. Mendeleyev’s initial discovery, made in the mid-nineteenth century, may have proven crucial to chemistry’s progress. Whenever chemical elements are arranged in this fashion, their properties follow a recurring pattern referred to as the “periodic rule,” which states that elements in the same column (group) have similar properties. The ordered arrangement of all of the elements and compounds in order of atomic number increasing is, the complete number of protons within the atomic nucleus-is referred to as the periodic table. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
